
Advancing Multiple Generations of Drug Candidates
Our strategy is to deliver on well-characterized psychedelic candidates and next generation candidates with enhanced drug profiles.
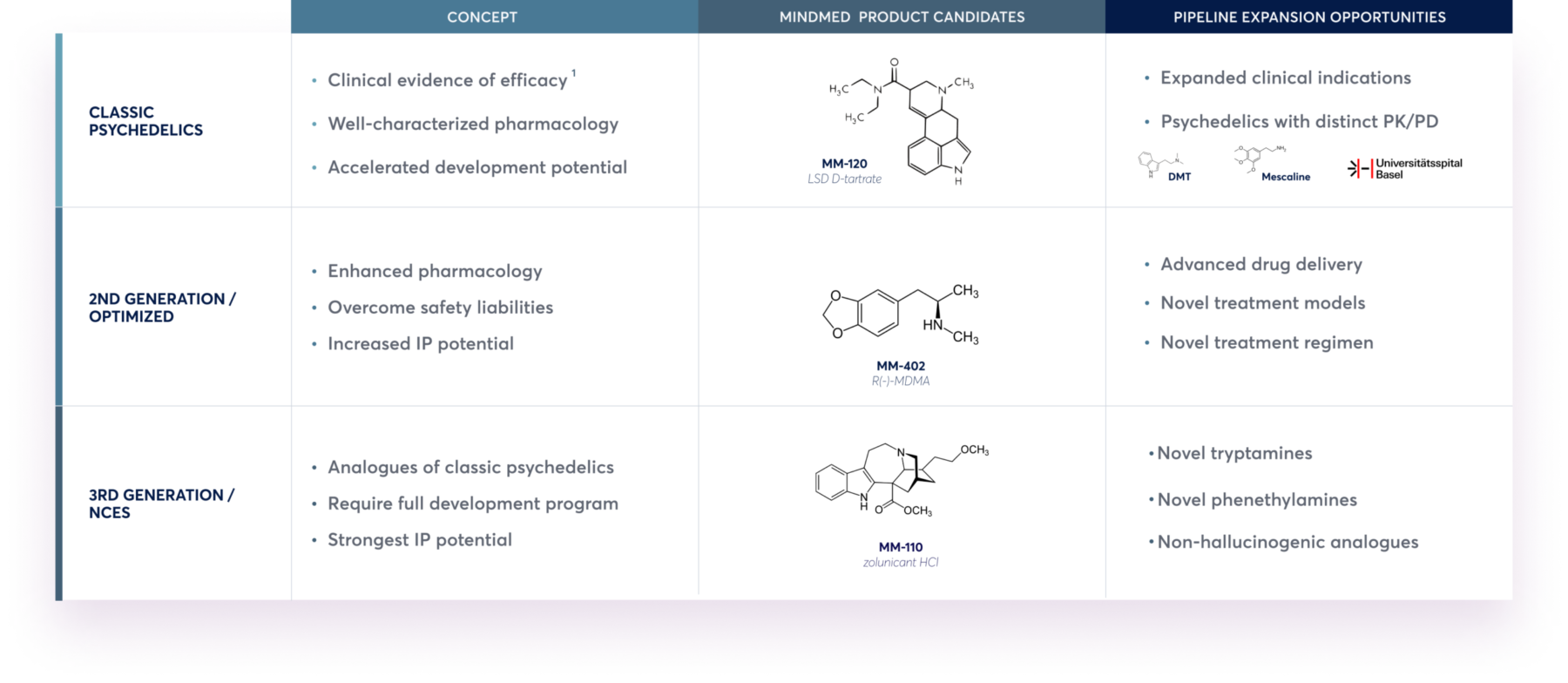
1. Gasser 2014; J. Nerv. Ment. Dis.; 202(7).
IP: intellectual property; DMT: N,N-dimethyltryptamine; MDMA: 3,4-methylenedioxymethamphetamine; NCE: new chemical entity; PD: pharmacodynamics; PK: pharmacokinetics